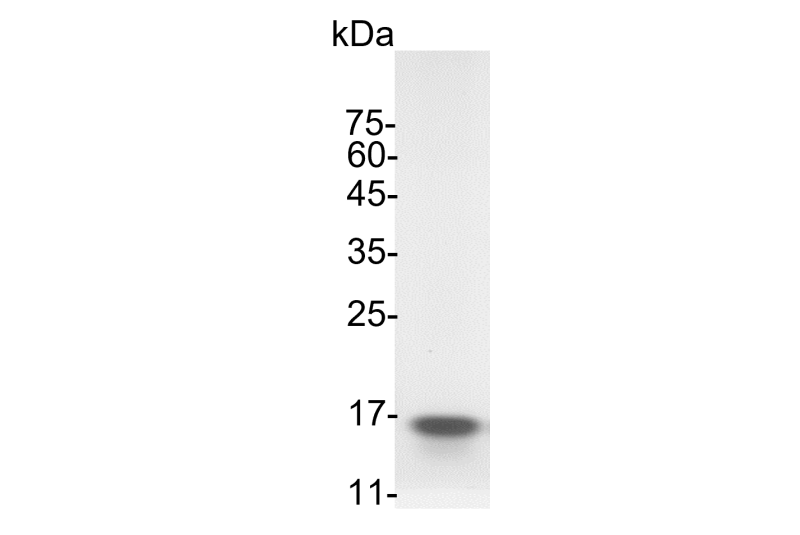GMP ® GM-CSF (Granulocyte-macrophage colony-stimulating factor), Human
Granulocyte-macrophage colony-stimulating factor (GM-CSF), also known as colony-stimulating factor 2 (CSF2), is a monomeric glycoprotein secreted by macrophages, T cells, mast cells, natural killer cells, endothelial cells and fibroblasts that functions as a cytokine. GM-CSF also plays a role in embryonic development by functioning as an embryokine produced by reproductive tract.
✔️ Over 98% purity
✔️ A potent immune stimulator
✔️ Ideal for a wide range of cell culture experiments, including dendritic cell differentiation, hematopoietic stem cell expansion, and more
✔️ Over 98% purity
✔️ A potent immune stimulator
✔️ Ideal for a wide range of cell culture experiments, including dendritic cell differentiation, hematopoietic stem cell expansion, and more
Sequence:
APARSPSPSTQPWEHVNAIQEARRLLNLSRDTAAEMNETVEVISEMFDLQEPTCLQTRLELYKQGLRGSLTKLKGPLTMMASHYKQHCPPTP
ETSCATQIITFESFKENLKDFLLVIPFDCWEPVQE with polyhistidine tag at the N-terminus
UnitProt ID:
P04141
Species of Origin:
Human
Expression System:
Escherichia coli
Endotoxin level:
<0.05 EU per 1 μg of the protein by the LAL method.
Activity:
Measure by its ability to induce TF-1 cells proliferation. The ED50 for this effect is<80 pg/mL.
The specific activity of recombinant human GM-CSF is approximately >1 x 107 IU/mg.
Purity:
>98% as determined by SDS-PAGE analysis.
Form:
Lyophilized
Storage Buffer:
Lyophilized from a 0.2 µm filtered solution of PBS, pH 8.0.
Reconstitution:
It is recommended to reconstitute the lyophilized protein in sterile H₂O to a concentration not less than 0.5 mg/mL and incubate the stock solution for at least 20 min to ensure sufficient re-dissolved.
Stability & Storage:
This product is stable after storage at:
• -20°C for 12 months in lyophilized state from date of receipt.
• -20°C or -80°C for 1 month under sterile conditions after reconstitution.
Avoid repeated freeze/thaw cycles.
Shipping conditions:
Blue ice
Background Information
Granulocyte-macrophage colony-stimulating factor (GM-CSF), also known as colony-stimulating factor 2 (CSF2), is a monomeric glycoprotein secreted by macrophages, T cells, mast cells, natural killer cells, endothelial cells and fibroblasts that functions as a cytokine. GM-CSF also plays a role in embryonic development by functioning as an embryokine produced by reproductive tract.
Quality Statement
Croyez GMP® recombinant proteins are manufactured in ISO 13485:2016 and GMP-certified facility.
The processes include:
● Animal-free reagent and laboratory
● Manufactured and tested under GMP guideline
● Testing and traceability of raw material
● Records of the maintenance and equipment calibration
● Personnel training records
● Batch-to-batch consistency
● Documentation of QA control and process changes
● Manufactured and tested under an ISO 13485:2016 certified quality management system
● Stability monitor of product shelf-life
Quality Assurance
At Croyez, we are committed to providing our valued customers with detailed product analytics to assist in their research endeavors. Our Certificate of Analysis includes the following lot-specific details:
● SDS-PAGE analysis and endotoxin level evaluation conducted on bulk QC lots.
● Lot-specific bioassay results ensuring compliance with established parameters, including microbial testing meeting USP <71> standards.
● Mycoplasma detection via PCR analysis.
We believe in transparency and strive to empower our customers with comprehensive information to aid in their decision-making process.
APARSPSPSTQPWEHVNAIQEARRLLNLSRDTAAEMNETVEVISEMFDLQEPTCLQTRLELYKQGLRGSLTKLKGPLTMMASHYKQHCPPTP
ETSCATQIITFESFKENLKDFLLVIPFDCWEPVQE with polyhistidine tag at the N-terminus
UnitProt ID:
P04141
Species of Origin:
Human
Expression System:
Escherichia coli
Endotoxin level:
<0.05 EU per 1 μg of the protein by the LAL method.
Activity:
Measure by its ability to induce TF-1 cells proliferation. The ED50 for this effect is<80 pg/mL.
The specific activity of recombinant human GM-CSF is approximately >1 x 107 IU/mg.
Purity:
>98% as determined by SDS-PAGE analysis.
Form:
Lyophilized
Storage Buffer:
Lyophilized from a 0.2 µm filtered solution of PBS, pH 8.0.
Reconstitution:
It is recommended to reconstitute the lyophilized protein in sterile H₂O to a concentration not less than 0.5 mg/mL and incubate the stock solution for at least 20 min to ensure sufficient re-dissolved.
Stability & Storage:
This product is stable after storage at:
• -20°C for 12 months in lyophilized state from date of receipt.
• -20°C or -80°C for 1 month under sterile conditions after reconstitution.
Avoid repeated freeze/thaw cycles.
Shipping conditions:
Blue ice
Background Information
Granulocyte-macrophage colony-stimulating factor (GM-CSF), also known as colony-stimulating factor 2 (CSF2), is a monomeric glycoprotein secreted by macrophages, T cells, mast cells, natural killer cells, endothelial cells and fibroblasts that functions as a cytokine. GM-CSF also plays a role in embryonic development by functioning as an embryokine produced by reproductive tract.
Quality Statement
Croyez GMP® recombinant proteins are manufactured in ISO 13485:2016 and GMP-certified facility.
The processes include:
● Animal-free reagent and laboratory
● Manufactured and tested under GMP guideline
● Testing and traceability of raw material
● Records of the maintenance and equipment calibration
● Personnel training records
● Batch-to-batch consistency
● Documentation of QA control and process changes
● Manufactured and tested under an ISO 13485:2016 certified quality management system
● Stability monitor of product shelf-life
Quality Assurance
At Croyez, we are committed to providing our valued customers with detailed product analytics to assist in their research endeavors. Our Certificate of Analysis includes the following lot-specific details:
● SDS-PAGE analysis and endotoxin level evaluation conducted on bulk QC lots.
● Lot-specific bioassay results ensuring compliance with established parameters, including microbial testing meeting USP <71> standards.
● Mycoplasma detection via PCR analysis.
We believe in transparency and strive to empower our customers with comprehensive information to aid in their decision-making process.
Reviews for GMP ® GM-CSF (Granulocyte-macrophage colony-stimulating factor), Human
Average Rating: 0 (0 Reviews )